Pharmacological Evaluation of Metal–Organic Frameworks (MOFs): Emerging Nanocarriers in Modern Pharmaceutics
DOI:
https://doi.org/10.64062/IJPCAT.Vol2.Issue1.4Keywords:
- Metal–Organic Frameworks (Mofs); Nanocarriers; Drug Delivery; Pharmacokinetics; Biodistribution; Nucleic Acid Delivery; Anticancer Therapy; Biomimetic Coatings; Stimuli-Responsive Systems; Theranostics; Pharmaceutical Nanotechnology
Abstract
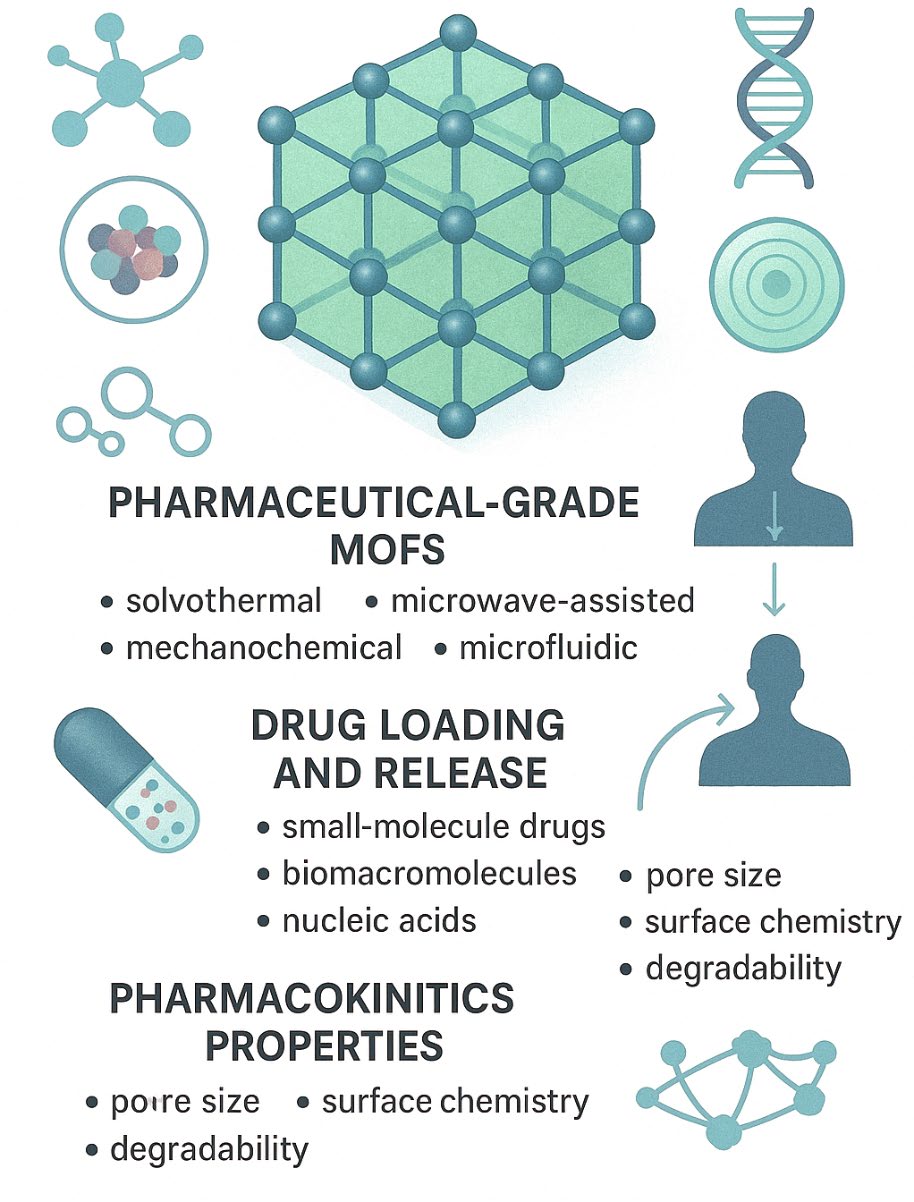
Metal–organic frameworks (MOFs) have emerged as powerful next-generation nanocarriers in modern pharmaceutics, offering unique advantages over conventional delivery systems through their exceptional porosity, tunability, and modular architecture. This review provides a comprehensive evaluation of MOF-based drug delivery platforms, emphasizing their structural fundamentals, synthesis strategies, drug-loading mechanisms, pharmacokinetics, and therapeutic potential. Pharmaceutical-grade MOFs are now engineered through advanced solvothermal, microwave-assisted, mechanochemical, and microfluidic methods, supported by green chemistry innovations and precise post-synthetic modifications. Their versatile loading mechanisms accommodate small-molecule drugs, biomacromolecules, and nucleic acids, while physicochemical attributes—such as pore size, surface chemistry, and degradability—dictate in vivo biodistribution and release kinetics. MOFs demonstrated significant pharmacological applications in cancer therapy, antimicrobial and antiviral treatment, gene delivery, gas storage, and enzyme-based therapeutics. However, concerns regarding cytotoxicity, degradation products, immunogenicity, and long-term accumulation highlight the importance of rigorous toxicological evaluation and biosafety-focused design. Emerging trends—including biomimetic coatings, MOF–polymer hybrids, stimuli-responsive systems, AI-guided design, and theranostic platforms—are rapidly advancing their clinical relevance. Despite ongoing challenges related to scalability, reproducibility, and regulatory approval, MOF nanocarriers hold strong potential to revolutionize personalized and precision medicine through smart, adaptable, and multifunctional therapeutic delivery systems
References
Downloads
Published
Issue
Section
License
Copyright (c) 2026 This is an Open Access article distributed under the terms of the Creative Commons Attribution (CC BY NC), which permits unrestricted use, distribution, and reproduction in any medium, as long as the original authors and source are cited. No permission is required from the authors or the publishers. (https://creativecommons.org/licenses/by-nc/4.0/)

This work is licensed under a Creative Commons Attribution 4.0 International License.





